 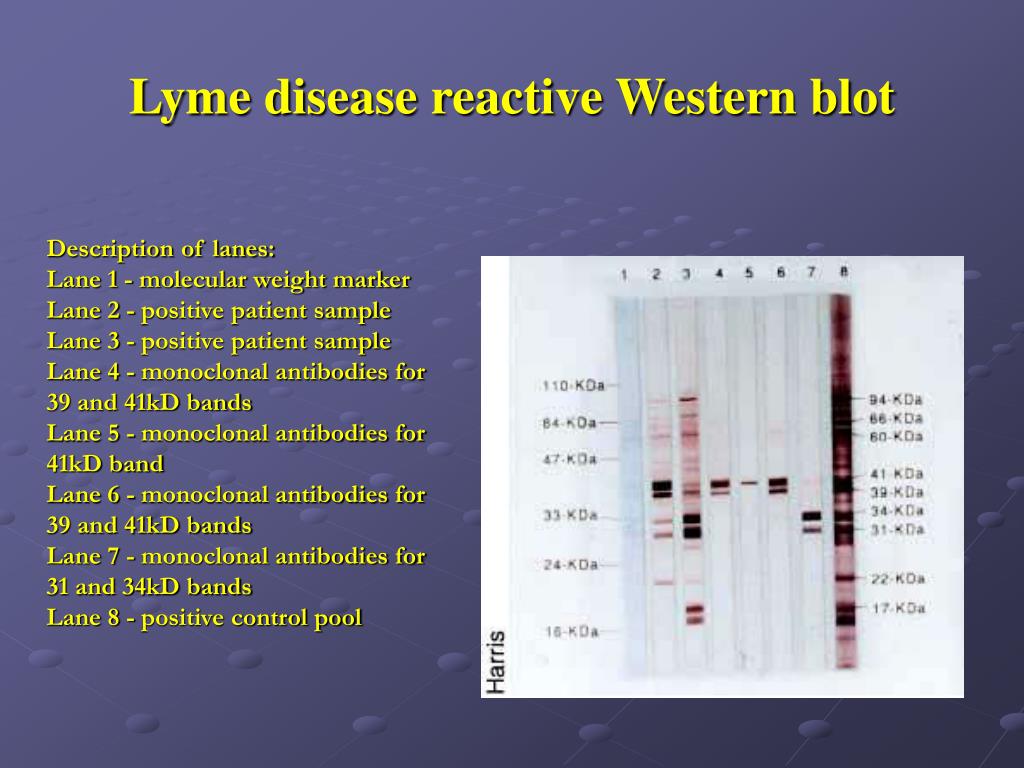
By conducting a Bradford assay, a colorimetric protein assay that exploits a dye’s interaction with protein, protein concentration is quantifiable. Intuitively, this is imperative for a valid experiment as unequal proteins per lane can skew the analysis. There must be an equal concentration of proteins per western blot sample. Collectively, this information underscores the need to tailor protein extraction to sample type and the target protein. 
PPIs are used to maintain the structure and phosphorylation status of the target protein from the activity of endogenous phosphatases upon cell lysis and exogenous phosphatases in the lysis microenvironment. As such, gentle buffers without detergents are required. Although rare, some antibodies will not be able to detect denatured samples. For example, radioimmunoprecipitation assay buffer (RIPA) is more adept for nuclear and mitochondrial proteins. Furthermore, the cell lysis buffer used in extraction should align with target protein cellular localization. 
For example, most tissue preparation is by homogenization or sonication however, osmotic shock or detergent lysis is more suited for easily lysed cells such as erythrocytes or cultured cells. There are numerous methods of extraction, and proper selection depends on the sample type. Western blot samples are first prepared by protein extraction with specialized cell lysis buffers and protease and phosphatase inhibitors (PPIs). Proper sample preparation for subsequent electrophoresis is crucial for downstream analysis. Donors with these patterns should undergo follow-up testing to document the presence or absence of HIV infection.Western blot relies on the principles of equal loading of proteins, separation of proteins by molecular weight, electrophoretic transfer to a suitable membrane, and probing of antibodies. Increased detection of env-only and p24/env-only WBs appears related to the enhanced sensitivity of newer enzyme immunoassays to gp41 and p24 antibodies. Env-only patterns predominated in 19, whereas p24/env-only patterns were more frequent following implementation of combined anti-HIV-1/HIV type 2 enzyme immunoassays in 1992.Ĭonclusion: Low-risk blood donors can have false-positive results on WB tests. The rate of detecting WBs with these false-positive patterns increased from 0.6 percent of all positive WBs from 1988 to 1990 (4/776) to 8 percent in 19 (52/683), and then it declined to 6 percent in 19 (47/783). For three of the four donors, env reactivity was mapped to a 20-amino acid N-terminal epitope of gp41. Env reactivity was restricted to nonglycosylated gp41 epitopes no gp120- specific reactivity was detected. Results: The four index-case donors denied risk and had stable WB reactivity during follow-up. WB records from American Red Cross blood centers were reviewed to determine the frequency of env- only and p24/env-only patterns, relative to all positive WBs, from 1988 through 1993. Index and/or follow-up specimens were tested by polymerase chain reaction (PCR), by overlapping recombinant env antigens and synthetic peptides in enzyme immunoassays, and by deglycosylated and denatured antigen WBs. Study Design and Methods: Four donors classified as WB- positive on the basis of env-only (3 cases) or p24/env-only (1 case) patterns were investigated. The present study was conducted to verify the existence of false-positive WBs among noninfected donors and to assess the extent of this problem. In a recent report, low-risk blood donors were identified in whom nonspecific reactivity to multiple env antigens in WB testing resulted in apparently false-positive WBs by these criteria. Background: The manufacturers' criteria for a positive human immunodeficiency virus type 1 (HIV-1) Western blot (WB) test were recently revised to require reactivity to only two of the following bands: p24, gp41, and gp120/160. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
